
There’s new hope for those seeking early detection of breast cancer.
Researchers, led by University College London, UK, have discovered a new marker, which could help diagnose fatal breast cancer up to one year earlier than current methods. They described it in a study published in the open access journal Genome Medicine this week.
The team discovered a region of DNA called EFC#93 showed abnormal patterns of DNA methylation in breast cancer samples. These are found in blood serum even before the cancer becomes detectable in the breast.
“For the first time, our study provides evidence that serum DNA methylation markers such as EFC#93 provide a highly specific indicator that could diagnose fatal breast cancers up to one year in advance of current diagnosis,” said Professor Martin Widschwendter, the study’s corresponding author. “This may enable individualized treatment, which could even begin in the absence of radiological evidence in the breast. We found the presence of EFC#93 DNA methylation in blood serum correctly identified 43 percent of women who went on to be diagnosed with fatal breast cancer within three to six months of giving serum samples, as well as 25 percent of women who went on to be diagnosed within six to twelve months of giving samples.”
DNA methylation is the addition of a methyl group to DNA, which often affects gene expression. Aberrant DNA methylation is common in human tumors and methylation changes occur quite early in breast cancer development.
Researchers first analyzed EFC#93 DNA methylation in blood serum samples from 419 breast cancer patients taken at two different times: after surgery (before the start of chemotherapy), and after completion of chemotherapy. They demonstrated that aberrant DNA methylation in samples taken before chemotherapy was a marker for poor prognosis independent of the presence of circulating tumor cells (cells that have shed from the primary tumor into the blood or lymphatic system and circulate throughout the body).
To find out whether EFC#93 can diagnose women with a poor prognosis before the cancer becomes detectable, researchers analyzed serum samples from 925 healthy women, 229 of whom went on to develop fatal and 231 of whom went on to develop non-fatal breast cancer, within the first three years of donating serum samples.
“The serum DNA methylation marker EFC#93 correctly identified 43 percent of women from serum tested six months in advance of their mammography-based breast cancer diagnosis who later died from the disease (sensitivity for fatal breast cancer) and also identified 88 percent of women who did not go on to develop breast cancer (specificity),” said Widschwendter. “Importantly, EFC#93 did not detect non-fatal breast cancers early. In comparison, mammography screening has a specificity of 88-92 percent but leads to very substantial over-diagnosis, which means that tumors are detected that would never have caused any clinical symptoms. Subject to further study, using cell-free DNA as a marker, as we have done here, is a promising way of avoiding this issue.”
A lack of appropriate serum samples is a key limitation of this kind of study. Blood samples that are not processed immediately after blood is drawn, or are not collected in special tubes, contain large amounts of normal ‘background’ DNA leaked by white blood cells, which makes it hard to detect small amounts of tumor DNA.
“The normal DNA in these samples usually emits a much stronger signal compared with the short fragments of tumor DNA,” said Widschwendter. “Yet despite the massive contamination of our population-based samples with normal DNA, we were able to observe a clear tumor DNA signal.”
Professor Widschwendter’s team is preparing for clinical trials in a large scale population-based cell-free DNA research program to see whether EFC#93 positive women, who do not have cancer that is detectable by mammography, would benefit from anti-hormonal therapy before the cancer becomes visible in the breast.
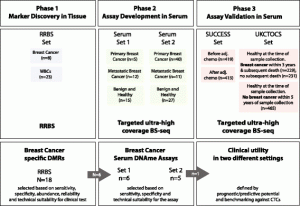
Study design. Using reduced representation bisulfite sequencing (RRBS), 31 human tissue samples were analyzed to identify a total of 18 regions which underwent thorough technical validation. Six regions were selected whose methylation status has been analyzed in two sets consisting of 110 serum samples. One marker (EFC#93) has been validated in two independent settings: (1) in SUCCESS study serum samples from BC patients before and after chemotherapy; and (2) in UKCTOCS serum samples from women before BC diagnosis (within three years) or who remained healthy for five years.

No comments yet.